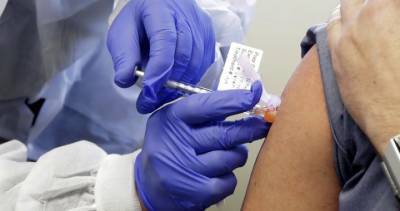
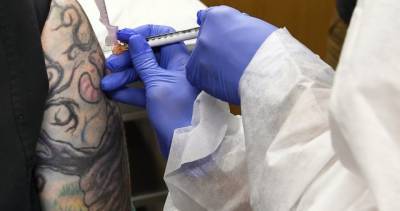
US set to approve Moderna vaccine
The US Food and Drug Administration (FDA) will "rapidly" work towards granting emergency approval of Moderna Inc's Covid-19 vaccine candidate, FDA Commissioner Stephen Hahn has said.
A panel of outside advisers to the FDA overwhelmingly endorsed the emergency use of Moderna's coronavirus vaccine. The committee voted 20-0 with one abstention that the benefits of the vaccine outweighed its risks in people aged 18 and older.
A week ago, the same panel backed a vaccine from Pfizer/BioNTech, leading to an FDA emergency use authorisation (EUA) a day later.
Following Mr Hahn's comments, the Financial Times reported that the FDA had decided to approve Moderna's vaccine candidate on an emergency basis.
Read more on rte.ie