NIH testing effectiveness, safety of mixing COVID-19 vaccines with potential boosters
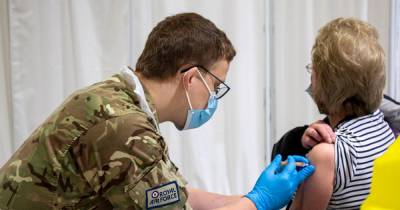
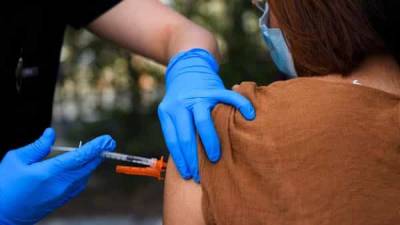
BETHESDA, Md. - The National Institute of Health has begun clinical trials for patients who have received both doses of the COVID-19 vaccine and giving them a different third booster shot to test the effectiveness and safety of mixing vaccines, the NIH announced on Monday.
For example, if a patient received both doses of the Moderna vaccine, the NIH is attempting to determine whether or not receiving a third booster shot of Pfizer will be just as effective and safe as receiving a booster of Moderna.
The trial is being funded by the NIH in correspondence with the Infectious Diseases Clinical Research Consortium, a network of clinical trials which cover the institute’s effort to test vaccines and treatments for deadly diseases like the novel.
Read more on fox29.com