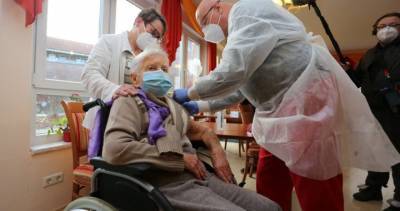
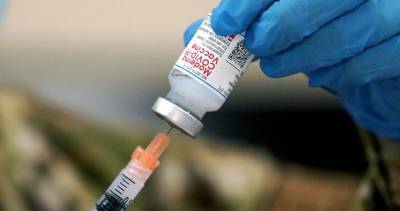
2nd COVID-19 vaccine by Moderna set for FDA approval in US after panel endorsement
In this photo illustration a medical syringe and a vial depicting fake coronavirus vaccine seen in front of the Moderna US biotechnology company logo. (Photo Illustration by Pavlo Gonchar/SOPA Images/LightRocket via Getty Images) WASHINGTON - A government advisory panel endorsed a second COVID-19 vaccine Thursday, paving the way for the shot to be added to the U.S.
vaccination campaign.The Food and Drug Administration is expected to follow the recommendation for the vaccine from Moderna and the National Institutes of Health.
The FDA advisers, in a 20-0 vote, agreed the benefits of the vaccine outweighed the risks for those 18 years old and up.The FDA's green light for emergency use is expected quickly.
Read more on fox29.com