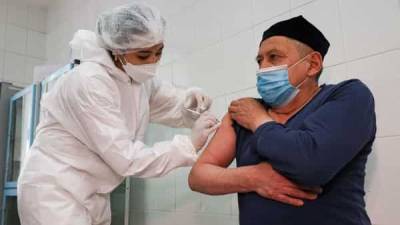
AstraZeneca lowers efficacy claim for COVID-19 vaccine, a bit, after board's rebuke
Jon CohenScience’s COVID-19 reporting is supported by the Heising-Simons Foundation.Seeking to quell a controversy of its own making, AstraZeneca yesterday issued new data from the latest clinical trial of its COVID-19 vaccine, slightly downgrading its previous estimate of how well the shots protect people from symptomatic disease.
The update came after an extraordinary rebuke issued late Monday night by the study's independent monitoring board, which complained that the company had used potentially misleading and “outdated” data in its initial analysis.The two-dose vaccine, made by AstraZeneca with technology developed by the University of Oxford, had 76% efficacy against symptomatic COVID-19 among the 32,000 trial participants in the.
Read more on sciencemag.org