CDC panel to discuss expanding COVID-19 booster shots to all US adults
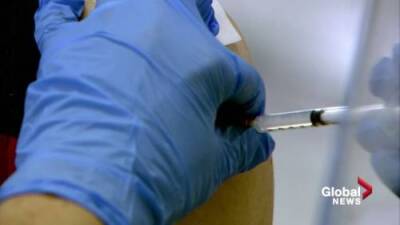
WASHINGTON - An influential advisory panel to the U.S. Centers for Disease Control and Prevention will meet on Friday to discuss expanding eligibility for COVID-19 vaccine booster shots to all American adults.
Both Pfizer and Moderna have asked U.S. regulators to allow boosters of its COVID-19 vaccine for anyone 18 or older. Pfizer submitted its application to the U.S.
Food and Drug Administration last week, and Moderna on Wednesday announced that it had filed an emergency use authorization request.The FDA was expected to sign off on at least Pfizer’s application before the advisory panel meets Friday.
Read more on fox29.com