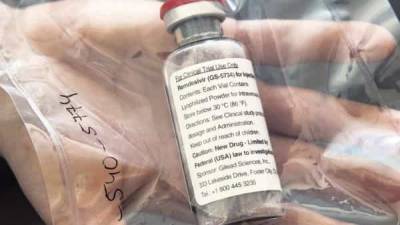
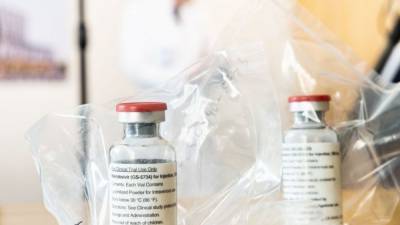
Cipla to sell anti-viral drug remdesivir as Cipremi in India
MUMBAI : Drug maker Cipla Limited on Sunday announced the launch of experimental anti-viral drug remdesivir under its brand name Cipremi.
Remdesivir is the only US FDA approved Emergency Use Authorisation (EUA) treatment for adult and paediatric patients hospitalized with suspected or laboratory confirmed COVID-19 infection.In May, US drug maker Gilead Sciences extended a voluntary non-exclusive license to Cipla to manufacture and market Cipla's generic version of remedisvir called Cipremi.The Drug Controller General of India (DCGI) has granted Cipla regulatory approval for restricted emergency use in the country as part of the accelerated approval process considering the urgent and unmet medical need, the company said in a statement.As.
Read more on livemint.com