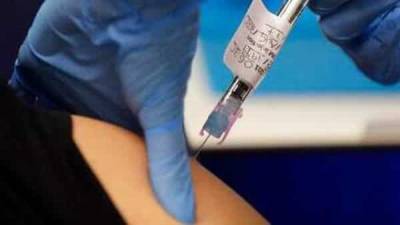
FDA chief says he could approve COVID-19 vaccine before completion of Phase 3 clinical trials
WASHINGTON - In an interview with the Financial Times, Food and Drug Administration Commissioner Dr. Stephen Hahn said a potential emergency authorization for a coronavirus vaccine before the completion of Phase 3 clinical trials would be “appropriate” if his agency found that the benefits outweighed the risks.“It is up to the sponsor [vaccine developer] to apply for authorization or approval, and we make an adjudication of their application,” Hahn said. “If they do that before the end of Phase 3, we may find that appropriate.
We may find that inappropriate, we will make a determination.”RELATED: People ‘in their 20s, 30s and 40s’ becoming key spreaders of COVID-19, experts sayHahn’s suggestion that the U.S.
Read more on fox29.com