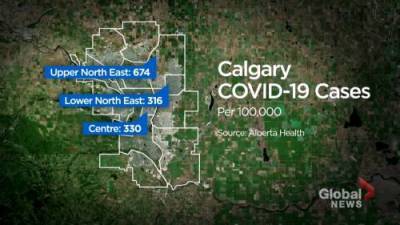
FDA to livestream public meeting on Pfizer-BioNTech potential COVID-19 vaccine
An illustration picture shows vials with Covid-19 Vaccine stickers attached, with the logo of US pharmaceutical company Pfizer, on November 17, 2020. (Photo by JUSTIN TALLIS/AFP via Getty Images) The U.S.
Food and Drug Administration has set a public meeting on Dec. 10 to discuss approving Pfizer and BioNTech’s experimental COVID-19 vaccine and will livestream it on YouTube, Facebook and Twitter.The companies asked the federal agency Friday for emergency use authorization after its research indicated that the vaccine candidate is 95% effective in preventing the coronavirus.
It’s the first vaccine request before the FDA that could serve as a major weapon in the fight to end the worsening global pandemic.“The FDA recognizes that transparency.
Read more on fox29.com