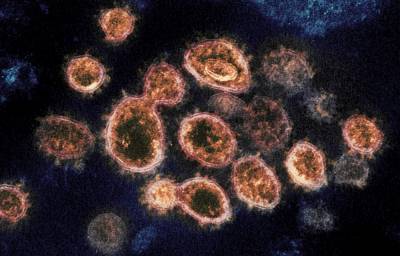
Officials reexamining AstraZeneca COVID vaccine data
The independent data and safety monitoring board (DSMB) overseeing the AstraZeneca vaccine trial notified the National Institute of Allergy and Infectious Diseases (NIAID) late yesterday that promising safety data may have "included outdated information from that trial, which may have provided an incomplete view of the efficacy data."In a statement, NIAID said it is urging AstraZeneca to work with the DSMB and provide the panel with the most recent efficacy data available.
Today, AstraZeneca responded that it would comply with the request. Fauci: AstraZeneca likely a very good vaccineAstraZeneca said the phase 3 US trial efficacy data were based on a specified interim analysis with a data cutoff of Feb 17 and that a preliminary review had
Read more on cidrap.umn.edu