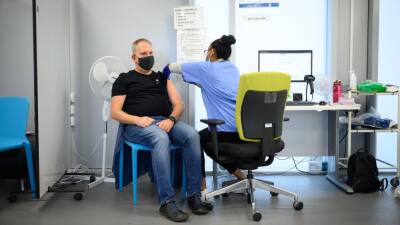
Pfizer asks FDA to authorize COVID-19 booster shot for all adults over age of 18
LOS ANGELES - On Tuesday, Pfizer and BioNTech announced they have submitted a request for emergency use authorization from the U.S.
Food and Drug Administration for a COVID-19 vaccine booster for all U.S. adults aged 18 and older.Currently, a booster is authorized by U.S.
regulators for individuals 65 years of age and older as well as adults with high risk of severe COVID-19. The companies said the request is based on results from a randomized Phase 3 trial with more than 10,000 participants in which they found boosters to have 95% effectiveness against symptomatic COVID-19 compared with the original two doses administered amid the prevalence of the highly transmissible delta variant.
Read more on fox29.com