Pfizer COVID vaccine gains full FDA approval
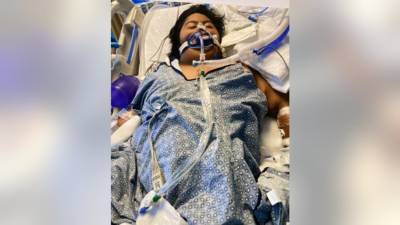
Earlier today the US Food and Drug Administration (FDA) granted the mRNA COVID-19 vaccine made by Pfizer-BioNTech full approval, a milestone in the ongoing pandemic.Though the vaccine has been used under an emergency use authorization (EUA) since last December, a full approval allows more businesses, schools, and organizations to mandate vaccine use among employees and students, and offers legitimacy to a vaccine some people believe has been rushed through safety evaluations and protocols."While this and other vaccines have met the FDA's rigorous, scientific standards for emergency use authorization, as the first FDA-approved COVID-19 vaccine, the public can be very confident that this vaccine meets the high standards for safety,
Read more on cidrap.umn.edu