Publix, Orlando-area COVID-19 vaccination sites modify operations as J&J use is paused
ORLANDO, Fla. – Across Central Florida, officials at COVID-19 vaccination sites scrambled to adjust operations as federal health authorities called for a temporary pause in the use of the Johnson & Johnson formula.
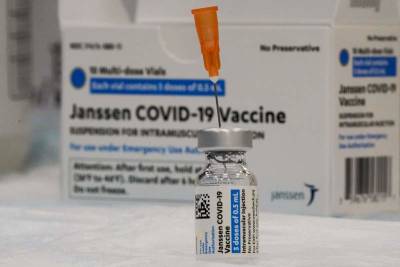
The move comes as six women, ages 18 to 48, reported potentially dangerous blood clots after receiving the single-dose J&J vaccine.
One woman died and a second woman in Nebraska has been hospitalized in critical condition, officials said. [TRENDING: Amber Alert for missing Fla.
girl | COVID symptoms persist year after man wakes from coma | Tornadoes hit Polk County] More than 6.8 million doses of the J&J vaccine have been administered in the U.S., the vast majority with no or mild side effects.
Read more on clickorlando.com