What to know about the Johnson & Johnson vaccine investigation
U.S. health officials paused use and distribution of Johnson & Johnson COVID-19 vaccines on Tuesday to investigate reports of potentially dangerous blood clots.
In a joint statement, the Centers for Disease Control and Prevention and the Food and Drug Administration said they were investigating unusual clots that occurred 6 to 13 days after vaccination.
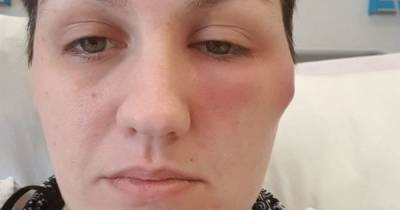
The clots occurred in veins that drain blood from the brain and occurred together with low platelets. All six cases were in women between the ages of 18 and 48; there was one death. [TRENDING: Amber Alert for missing Fla.
girl | COVID symptoms persist year after man wakes from coma | Tornadoes hit Polk County] Here are key takeaways about the vaccine investigation: How long will J&J shots be
Read more on clickorlando.com