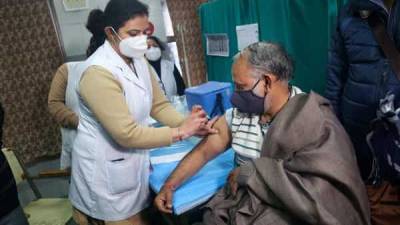
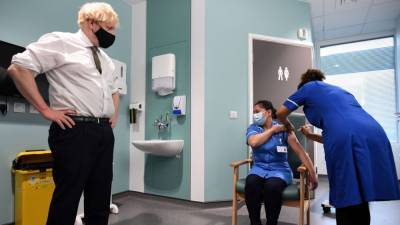
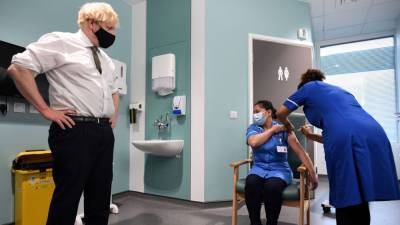
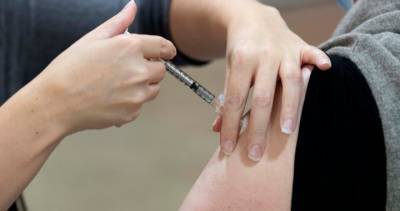
AstraZeneca and Oxford’s bumpy partnership hangs over covid vaccine’s future
Also Read | The march of 2020 in 10 key long readsThe partners have promised to distribute their vaccine in huge quantities and sell it cheaply throughout the pandemic.
In the U.S., AstraZeneca expects to have data from a large American clinical trial to show to the Food and Drug Administration by February, said a person familiar with the trial.The partners hope for authorization across Europe by February, and a green light in Australia could also come soon, this person said.
AstraZeneca’s manufacturing partner in India has asked for regulatory authorization there. All depend on regulatory sign-off.Complicating those plans are doubts seeded by the partners’ own handling of their ambitious project.
Read more on livemint.com