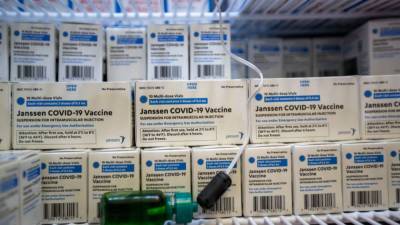
CDC advisory panel to hold emergency meeting Wednesday over J&J COVID-19 vaccine
WASHINGTON - The U.S. Center for Disease Control and Prevention’s advisory panel will meet Wednesday to discuss reports of rare but severe blood clots among a handful of people who received the Johnson & Johnson COVID-19 vaccine.The Advisory Committee on Immunization Practices (ACIP) is scheduled to review data on six cases of clotting in the United States.
All cases were in women between the ages of 18 and 48, who experienced symptoms roughly six to 13 days after getting the shot.
Officials said one of the patients had died and another was in critical condition.Nearly 7 million J&J doses have been administered in the U.S.
Read more on fox29.com