Fast-track applications for covid jabs a worry
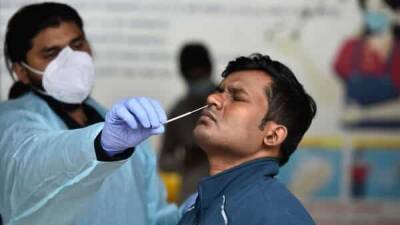
Serum Institute of India (SII) and Bharat Biotech International Ltd have raised concerns over insufficient clinical trial data in the public domain, experts said.Unlike foreign vaccine manufacturers, which first made their data public and then applied for emergency licences, both SII and Bharat Biotech submitted their applications to Drug Controller General of India (DCGI) V.G.
Somani even before completing their phase III clinical trials.In fact, Bharat Biotech began phase III enrolment for its vaccine candidate just 25 days ago and is using its phase I and II safety and immunogenicity data for seeking emergency licensure.“The regulator has to be satisfied with the risk-benefit ratio, and in this situation, if the benefit is more, he.
Read more on livemint.com