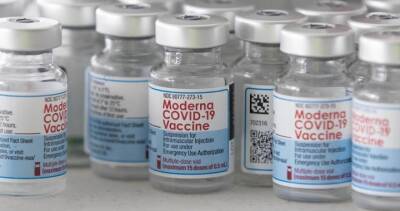
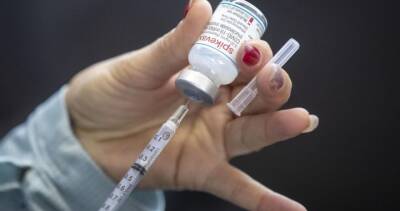
Serum Institute expects India nod for Covid-19 vaccine emergency use in next few days: Report
Serum Institute of India, which is also manufacturing the Oxford-AstraZeneca coronavirus vaccine in India, said on Monday it expected the government to approve the shot for emergency use in a few days, reported Reuters.Serum Chief Executive Adar Poonawalla also told reporters that the company had already made 40 million to 50 million doses of the vaccine.According to a Mint report, India is likely to approve Oxford-AstraZeneca's coronavirus vaccine for emergency use by next week after its local manufacturer submitted additional data sought by authorities.This could be the first country to give the regulatory green light for the British drugmaker's vaccine as the British medicine regulator continues to examine data from the trials.Currently,.
Read more on livemint.com