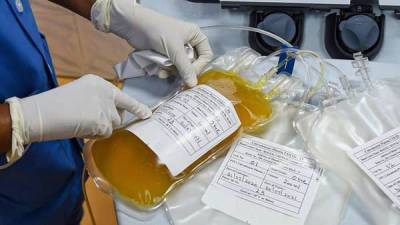
In FDA’s green light for treating COVID-19 with plasma, critics see thin evidence—and politics
Kai Kupferschmidt, Jon CohenScience’s COVID-19 reporting is supported by the Pulitzer Center and the Heising-Simons Foundation.At a highly unusual Sunday night press conference, U.S.
President Donald Trump revealed what he described as “a very historic breakthrough” in the fight against COVID-19 that “would save countless lives”: The U.S.
Food and Drug Administration (FDA) had issued an Emergency Use Authorization (EUA) for convalescent plasma to treat people with severe COVID-19.The authorization could allow more hospitalized patients to receive the antibody-rich plasma, which is donated by people who have recovered from the disease.
Read more on sciencemag.org