Interferon beta-1a may help COVID-19 patients recover faster
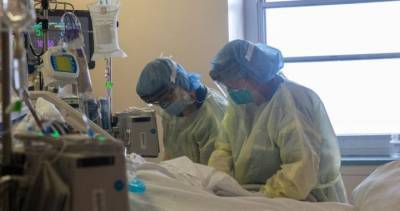
The Lancet Respiratory Medicine yesterday published the results of a clinical trial showing that the inhaled immune response protein interferon beta-1a reduced the chances of hospitalized COVID-19 patients becoming severely ill or dying of their infection.The double-blind, randomized, controlled trial, conducted at nine hospitals in the United Kingdom, involved 98 patients recruited from Mar 30 to May 30.
The patients were randomly assigned to receive either the inhaled interferon beta-1a formulation SNG001 or a placebo once daily for as long as 14 days.The patients who received SNG001 were twice as likely to improve clinically at day 15 or 16 than those receiving a placebo (21 of 48 [44%] vs 11 of 50 [22%]).
Read more on cidrap.umn.edu