Intranasal covid-19 vaccine safe, immunogenic: Bharat Biotech
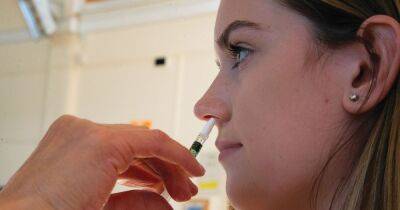
Bharat Biotech International Ltd. on Monday said its intra-nasal covid-19 vaccine, BBV154, is safe, well-tolerated, and immunogenic in subjects studied during phase 3 clinical trials for primary dose and a heterologous booster dose.
The company said data from phase 3 trials have been submitted for approval to national regulatory authorities. BBV154 is a recombinant replication-deficient adenovirus vectored vaccine with a pre-fusion stabilised spike protein.
The vaccine candidate was evaluated earlier in phase I and II clinical trials with successful results. BBV154 has been specifically formulated to allow intra-nasal delivery.
In addition, the nasal delivery system has been designed and developed to be cost-effective in low and middle-income countries.
Read more on livemint.com