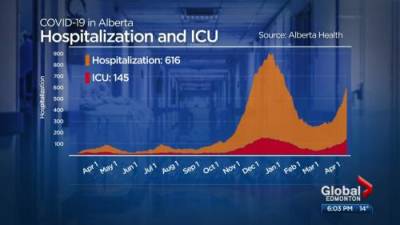
Johnson & Johnson files application for single-shot covid vaccine in India: Report
COVID-19 of the Central Drugs Standard Control Organisation (CDSCO) to take a decision on its application.This comes close to the heels of the central government last week deciding to fast-track emergency approvals for all foreign produced coronavirus jabs that have been given a similar nod by the World Health Organization or regulators in the United States, Europe, Britain or Japan.Such vaccines will be given emergency use approval mandating the requirement of post-approval parallel bridging clinical trial in place of conduct of local clinical trial under the provisions of the New Drugs and Clinical Trials Rules 2019, the health ministry had said.According to sources, Johnson & Johnson had applied on April 12 in the Global Clinical Trial.
Read more on livemint.com