US ready to distribute J&J COVID vaccine, White House says
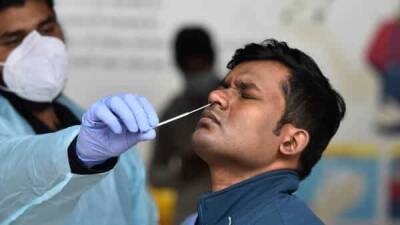
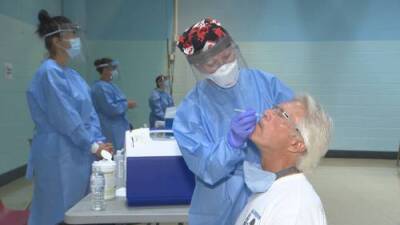
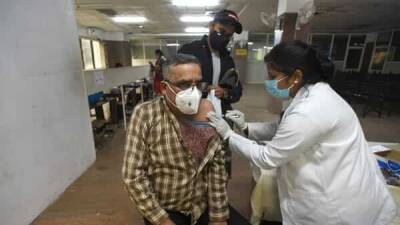
Today leaders of the White House COVID-19 response team said they are ready to distribute Johnson & Johnson's COVID-19 vaccine when and if the Food and Drug Administration (FDA) issues an emergency use authorization (EUA)—which could come as early as this weekend."We are doing the work so if the EUA is granted we will waste no time in getting life-saving vaccines into the arms of Americans," said Jeff Zients, the Biden administration's COVID-19 czar during today’s press conference on response efforts.Zients said governors have received information about how to possibly distribute the Johnson & Johnson vaccine, which, unlike the current approved and in-use vaccines, requires only one shot and standard refrigeration.If approved, the 3 million
Read more on cidrap.umn.edu