FDA grants EUA for Gilead’s remdesivir to treat Covid-19
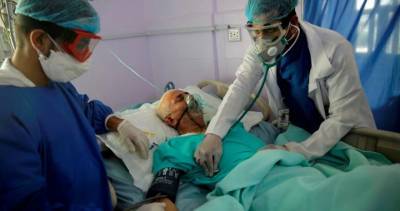
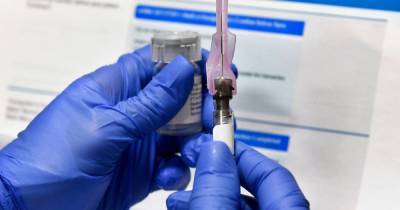
Sign up here for GlobalData's free bi-weekly Covid-19 report on the latest information your industry needs to know.The US Food and Drug Administration (FDA) has granted emergency use authorisation (EUA) for Gilead Sciences’ Veklury (remdesivir) to treat all hospitalised patients with Covid-19.
Previously, the drug received emergency authorisation for treating patients hospitalised with severe form of the disease. Veklury is an experimental nucleotide analogue with broad-spectrum antiviral activity in-vitro and also in-vivo in animal models against different emerging viral pathogens.
The drug is being studied in Phase III clinical trials globally in various Covid-19 patient populations, formulations and as a combination with other therapies..
Read more on pharmaceutical-technology.com