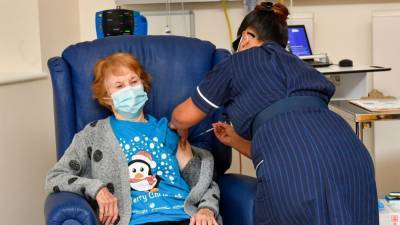
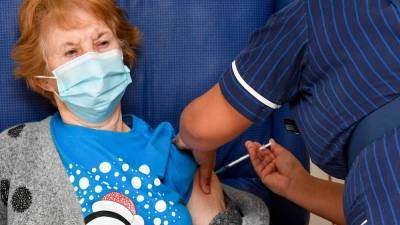
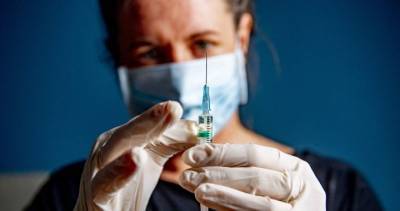
Strict tests done, no corners cut to approve Pfizer-BioNTech Covid vaccine: UK regulator
Pfizer-BioNTech's Covid-19 vaccine for general use next week.June Raine, chief executive of the Medicines and Healthcare products Regulatory Agency (MHRA) said it had used overlapping trials and "rolling reviews" to reach the determination.ALSO READ: India can’t make up its mind on PSUs, 30 years after liberalisationEarlier today, British officials authorized Pfizer-BioNTech's Covid-19 vaccine candidate for emergency use, greenlighting the world’s first shot against the virus that’s backed by rigorous science and taking a major step toward eventually ending the pandemic."This vaccine has only been approved because those strict tests have been done and complied with.
Read more on livemint.com