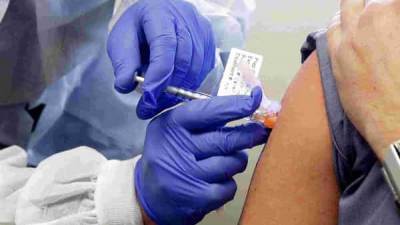
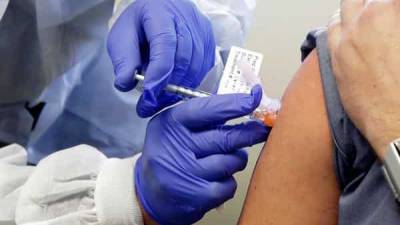
US aims to acquire 1 million doses of COVID-19 antibody by the end of the year
COVID-19 in anticipation of regulators authorizing their emergency use, federal health officials said on Friday.The government expects to have 1 million doses of the so-called monoclonal antibody treatments on hand before year-end.The treatments have come into the spotlight after President Donald Trump received one such experimental therapy last week for his case of COVID-19.Eli Lilly & Co.
and Regeneron Pharmaceuticals Inc. this week asked the US Food and Drug Administration for emergency-use authorizations for their antibody therapies but haven’t yet received clearance.
Trump has since repeatedly promised to hasten their approval, widen access and provide them to Americans for free.“We have engaged in a number of contracts for procurement.
Read more on livemint.com