US halts J&J COVID vaccine after reports of blood clots
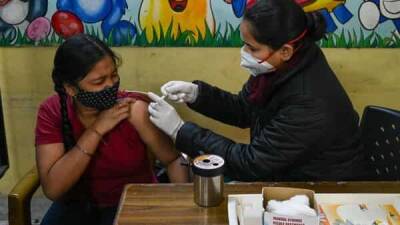
The US Food and Drug Administration (FDA) and the Centers for Disease Control and Prevention (CDC) this morning said the United States should pause the administration of the Johnson & Johnson COVID-19 vaccine immediately, while the agencies examine how and if the vaccine was linked to six cases of a rare type of blood clots in women 18 to 49, including one death."Right now I'd like to stress these events seem to be extremely rare," said Janet Woodcock, MD, acting FDA commissioner.
Approximately 7 million doses of the Johnson & Johnson, or J&J, vaccine have been administered in the United States.Peter Marks, MD, director of the FDA's Center for Biologics Evaluation and Research, said people who recently got the J&J vaccine should not panic,
Read more on cidrap.umn.edu