US HHS expedites development and manufacture of Covid-19 vaccines
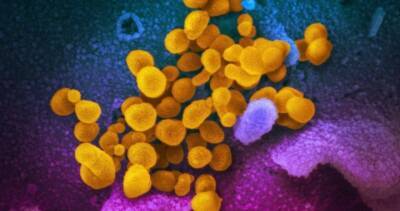
The US Department of Health and Human Services (HHS) is accelerating the development and manufacture of vaccines against the Covid-19 coronavirus infection.
HHS collaborated with Johnson & Johnson (J&J) unit Janssen Research & Development (R&D) and biotechnology company Moderna to advance vaccine candidates.
The Biomedical Advanced Research and Development Authority (BARDA) at HHS agreed to support non-clinical studies and a Phase I trial of Janssen’s vaccine candidate, Ad26 SARS-CoV-2, for Covid-19.
In the clinical trial, the vaccine will be assessed for its safety and ability to generate an immune response in healthy participants.
Read more on pharmaceutical-technology.com