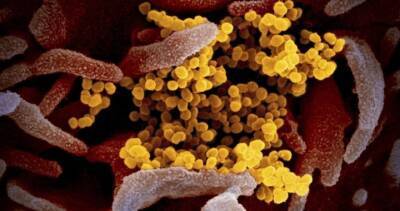
Brazil halts trials of Chinese Covid-19 vaccine
Brazil's health regulator has said it had suspended clinical trials of a Chinese-developed Covid-19 vaccine after an "adverse incident" involving a volunteer recipient.
The setback for CoronaVac, developed by Chinese pharmaceutical firm Sinovac Biotech, comes as US pharmaceutical giant Pfizer said yesterday its own vaccine candidate had shown 90% effectiveness, sending global markets soaring and raising hopes of an end to the pandemic.
The Brazilian regulator, Anvisa, said in a statement it had "ruled to interrupt the clinical study of the CoronaVac vaccine after a serious adverse incident" on 29 October.
It said it could not give details on what happened because of privacy regulations, but that such incidents included death, potentially
Read more on rte.ie