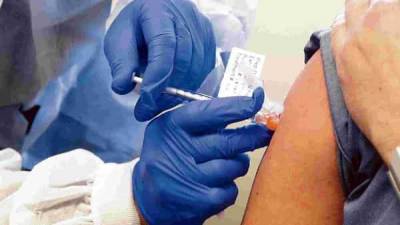
Covaxin: Behind the scenes of the Covid-19 vaccine trials
New Delhi: The moment Kumara Prashadh saw a post by a Chennai doctor on Twitter in August, requesting people to volunteer for the phase II clinical trials of covaxin, he knew he had to participate.
Like much of the world, the IT employee was closely following news about the race to develop a vaccine for the novel coronavirus.
Since covaxin was India’s indigenous covid-19 vaccine, Prashadh considered it his duty as a citizen, but he’d also read about a shortage of volunteers in the UK for vaccine trials. “I didn’t want that to happen in my country.
Read more on livemint.com