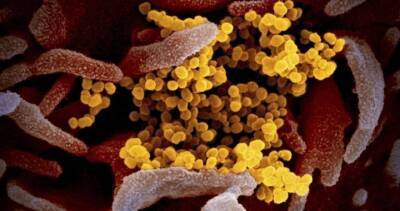
Novavax sees start of COVID vaccine trial in US in coming weeks after second delay
New York: Vaccine maker Novavax Inc said on Monday it has pushed back the start of a U.S.-based, late-stage trial for its experimental COVID-19 vaccine and now expects it to begin in the coming weeks instead of November.It is the second time that Novavax, which already has a late-stage UK trial underway, has rescheduled the Phase 3 trial after first flagging an October start, hampered by issues in scaling up its manufacturing.Shares of the U.S.-based company, which lags behind larger rivals Pfizer Inc and Moderna Inc in their COVID-19 vaccine development timelines, fell 6% on the latest delay.Novavax plans to use vaccine material produced at commercial scale for the trial in the United States and Mexico and has been working closely with the.
Read more on livemint.com