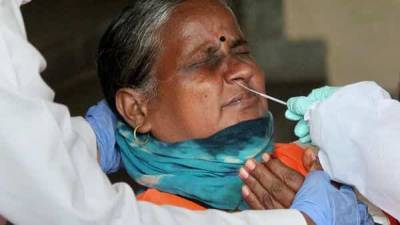
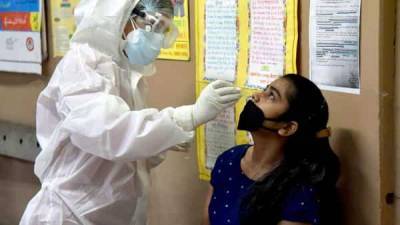
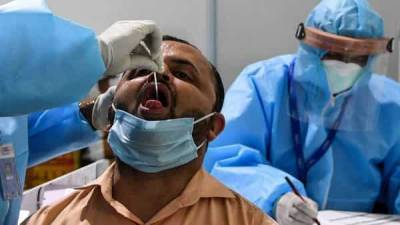
Oxford COVID-19 vaccine: Panel recommends DCGI to grant Serum Institute approval for human trial
New Delhi: A subject expert committee on COVID-19 on Friday recommended to the country's drug regulator that permission be granted to Serum Institute of India (SII) for conducting the second and third phases of human clinical trials of the Oxford vaccine candidate.The recommendations of the committee at the Central Drugs Standard Control Organisation (CDSCO) have been sent to the Drugs Controller General of India (DCGI) for approval, highly-placed sources told PTI.They said that the SII had submitted a revised proposal on Wednesday after the expert panel on Tuesday, following deliberations over its application, had asked it to revise its protocol for the trials besides seeking some additional information."An urgent meeting was held by the.
Read more on livemint.com