AstraZeneca data from U.S. vaccine trial may be outdated, incomplete: health agency
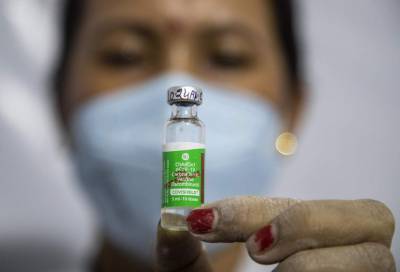
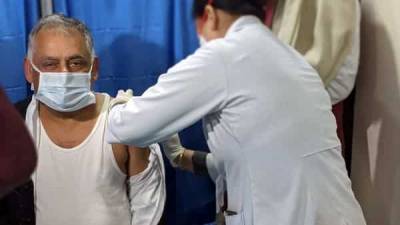
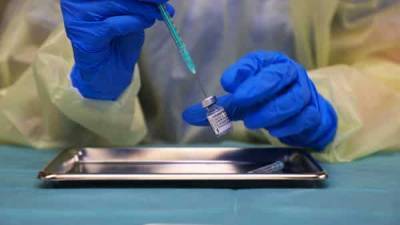
AstraZeneca has been informed that it may have provided outdated data on the efficacy of its COVID-19 vaccine from a large-scale clinical trial in the U.S., the country’s National Institute of Allergy and Infectious Diseases (NIAID) said on Tuesday.In an early morning statement, the NIAID said it had been informed by the national Data and Safety Monitoring Board (DSMB) late Monday that it was “concerned” about the information AstraZeneca provided.
Read more on globalnews.ca