BARDA head says he was pushed out after questioning malaria drugs for COVID-19
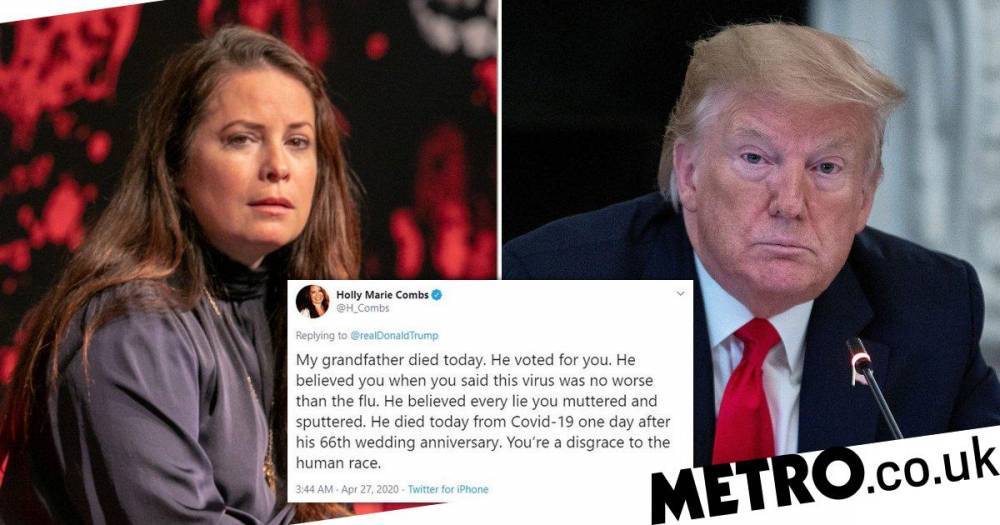
Rick Bright, MD, the former director of the Biomedical Advanced Research and Development Authority (BARDA), said he was pushed out of his position earlier this week because he insisted government investment in COVID-19 research should go to safe and scientifically vetted solutions, and not unproven drugs, especially the antimalarials championed by President Donald Trump.Bright, via his lawyer, issued a statement on his demotion late yesterday evening."Specifically, and contrary to misguided directives, I limited the broad use of chloroquine and hydroxychloroquine, prompted by the Administration as a panacea, but which clearly lack scientific merit," Bright said. "I rightly resisted efforts to provide an unproven drug on demand to the
Read more on cidrap.umn.edu