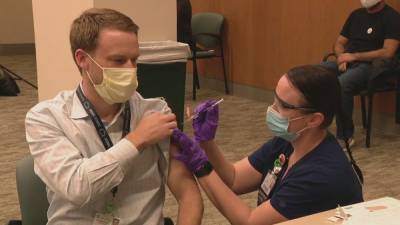
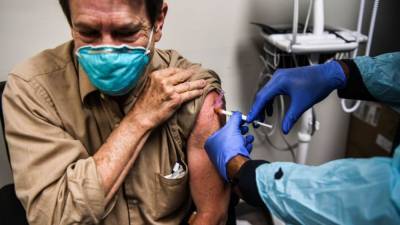
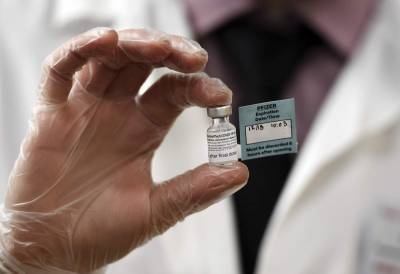
European regulators to review Pfizer Covid vaccine next week
Pfizer Inc. and BioNTech SE’s Covid-19 vaccine next week, earlier than initially planned, amid growing pressure across the continent to speed up approval of the shot.A European Medicines Agency committee will meet Monday to consider the vaccine after receiving more data from the developers, the drugs watchdog said in a statement Tuesday.
Once a recommendation is made, the European Commission is expected to decide whether to clear the shot within days.Also Read: How rural schooling is going into the darkLeaders in Germany, Italy and Poland, among other countries, have been pushing the European Union regulator to speed up its review as vaccination campaigns begin for high-risk people in the U.K.
Read more on livemint.com