GAO highlights COVID vaccine supply chain, drug transparency issues
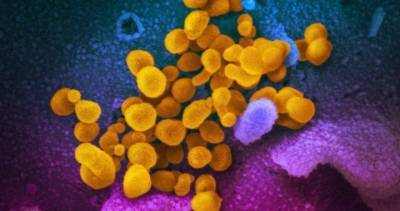
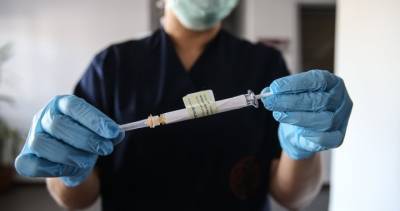
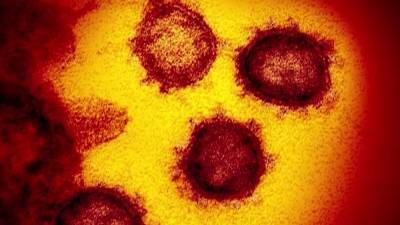
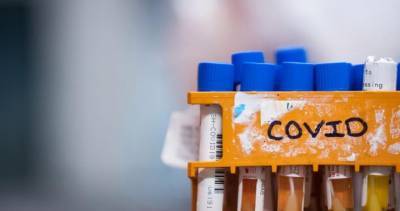
A report released yesterday by the US Government Accountability Office (GAO) says supply chain issues could interfere with a smooth rollout of a COVID-19 vaccine and that the Food and Drug Administration (FDA) should disclose scientific review of vaccine and drug efficacy and safety data when issuing emergency use authorizations (EUAs).The report states that Operation Warp Speed, an effort of the Department of Health and Human Services, the Department of Defense, and private industry to dramatically accelerate development of COVID-19 vaccines and therapeutics, has condensed the timeline by directing more than $10 billion to vaccine manufacturers.
Read more on cidrap.umn.edu