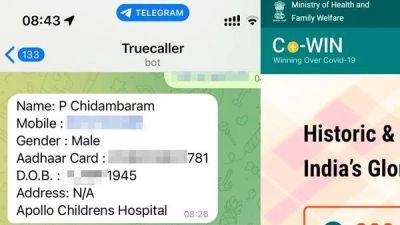
Govt bans 14 fixed dose combination drugs including Nimesulide and Paracetamol, citing health risk; check full list here
FDC drugs are those which contain a combination of two or more active pharmaceutical ingredients (APIs) in a fixed ratio. These banned drugs included those used for treating common infections, cough and fever combinations such as: Nimesulide Paracetamol dispersible tablets, Chlopheniramine Maleate Codeine Syrup, Pholcodine Promethazine, Amoxicillin Bromhexine and Bromhexine Dextromethorphan Ammonium Chloride Menthol, Paracetamol Bromhexine Phenylephrine Chlorpheniramine Guaiphenesin and Salbutamol Bromhexine.
The expert committee said that there is "no therapeutic justification for this FDC (fixed dose combination) and the FDC may involve risk to human beings.
Hence, in the larger public interest, it is necessary to prohibit the manufacture, sale or distribution of this FDC under section 26 A of the Drugs and Cosmetics Act, 1940.
In view of the above, any kind of regulation or restriction to allow for any use in patients is not justifiable." As per the notification issued by the Health Ministry, "And whereas, on the basis of the recommendations of the Expert Committee and the Drugs Technical Advisory Board, the Central Government is satisfied that it is necessary and expedient in public interest to regulate by way of prohibition the manufacture for sale, sale and distribution for human use of the said drug in the country," the notification stated.
Read more on livemint.com