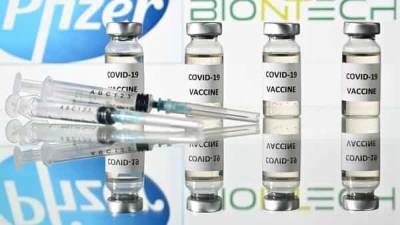
Pfizer/BioNTech seek first Covid-19 vaccine approval in US
Pfizer and its German partner BioNTech have formally submitted an application to the US Food and Drug Administration to authorise their Covid-19 vaccine for emergency use.
Pfizer's Chief executive Dr Albert Bourla said it is a critical milestone in their journey to deliver the vaccine to the world.
The companies have also begun regulatory submissions in the European Union, the UK, Australia, Canada and Japan. The FDA said its vaccines committee would meet on 10 December to discuss the request for emergency use authorisation.
If approved it could be in use by mid-December. "The FDA recognises that transparency and dialogue are critical for the public to have confidence in Covid-19 vaccines," the organisation's head Stephen Hahn said in a
Read more on rte.ie