EXPLAINER: Final steps in US review of COVID-19 vaccine
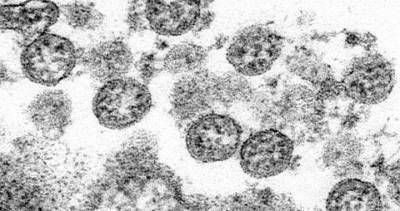
WASHINGTON – Pfizer’s COVID-19 vaccine is entering the final phase of review before the U.S. government decides whether to allow millions to get the shots.The Food and Drug Administration posted a positive review of the Pfizer vaccine on Tuesday and will hold a public hearing on Thursday.
Next week, it will do the same thing for Moderna's coronavirus vaccine candidate.A look at the process:FDA REVIEWThe agency's scientific review is a key step — not just for the U.S. — but for countries around the world weighing whether to begin using a vaccine.
Read more on clickorlando.com