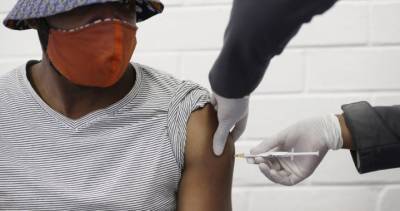
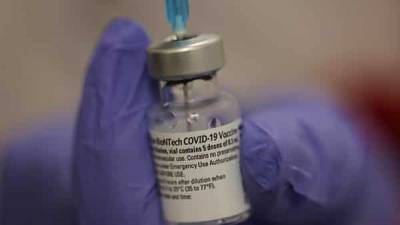
India readies 60 crore Covid-19 vaccine jabs with vast election machinery: Report
SUPPLIES AND STORAGEAt present, Indian regulators are considering three vaccines for emergency use authorisation, including those from Pfizer Inc, AstraZeneca and Bharat Biotech.But, Pfizer's limited stockpiles and its extreme storage condition requirements at minus 70 degrees Celsius or below, would likely limit its use in India, which lacks such infrastructure."In a theoretical scenario, where there is no vaccine with conventional cold chain requirement, minus 70 degrees Celsius capacities will have to be created, and we will do so," Paul said.Paul said the government is also in talks with Moderna, which also has requirements for ultra-cold storage.India does not anticipate supplies from either Pfizer or Moderna until the second half of.
Read more on livemint.com