3 COVID-19 tests recalled after FDA warns about ‘high risk’ of false results
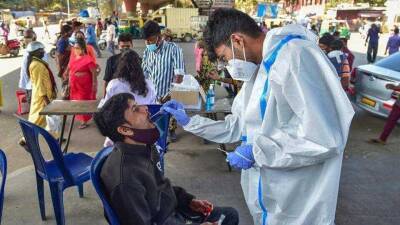
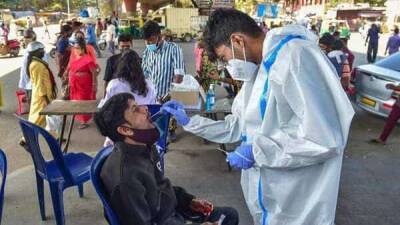
A Walmart worker uses a nasal swab in one of the new government-issued COVID-19 Antigen Rapid test kits she received as she self-tests while at home on February 8, 2022 in Provo, Utah. (Photo by George Frey/Getty Images) The maker of a certain brand of COVID-19 tests has issued a recall because the tests are not authorized, cleared or approved by the U.S.
Food and Drug Administration (FDA).LuSys Laboratories recalled three test types: a saliva antigen test, a nasal antigen test and an IgG/IgM antibody test.
These tests were at the center of an FDA safety memo earlier this year, in which the agency warned people to stop using LuSys Laboratories’ tests because there was likely a high risk of false results.
The FDA said the company did not provide appropriate validation data to show that the tests can perform accurately, meaning "there is a risk of potential false negative, false positive, and misinterpretation of results from these tests."RELATED: 2nd round of free at-home COVID tests available now, White House saysThe FDA believes these tests were distributed for use in laboratories and for at-home testing.
Read more on fox29.com