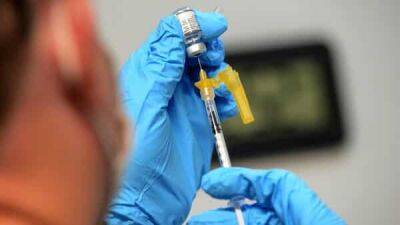
Pfizer files for emergency use of its BA.4/BA.5 Omicron booster
As the nation braces for another possible spike in COVID-19 activity, Pfizer and BioNTech today announced that they have submitted an application to the US Food and Drug Administration (FDA) for emergency use of its bivalent (two-strain) booster that targets the BA.4 and BA.5 Omicron variants.Shipping could begin in SeptembermRNA vaccine makers have been working on bivalent booster shots targeting the original Omicron variant, but in late June FDA vaccine advisers recommended that the boosters include the more recent BA.4 and BA.5 subvariants.
The more transmissible viruses, also known to evade earlier immunity from vaccines and previous infection, have fueled outbreaks in several countries and led to persistently high levels in many US communities over the summer.The nation's COVID-19 activity has been at a stubbornly elevated level this summer, and officials worry about more surges as schools resume and people gather indoors as the weather cools.In a statement, Pfizer said its EUA would clear the booster for use in people ages 12 and older.The company said the clinical trial is expected to begin later this month, but it has already scaled up production and will be ready to ship the vaccine in September, as soon as the FDA approves the emergency use authorization (EUA) application.To speed the consideration of the EUA, the FDA told Pfizer it could submit clinical data for its bivalent BA.1 vaccine alongside preclinical and manufacturing data on its BA.4/BA.5 vaccine.
Preclinical data in animals suggest that the BA.4/BA.5 bivalent booster prompts a strong neutralization antibody response against Omicron BA.1, BA.2, and BA.4/BA.5 variants, as well as to the original SARS-CoV-2 virus.Pfizer also said it will complete a
Read more on cidrap.umn.edu